In Fruit Fly Brain, A Ring for Navigation
If you slowly turn around with your eyes closed, chances are you’ll be able to keep track of which direction you’re facing. Neurons known as head-direction cells use a combination of past sensory knowledge and your own movement to calculate where you are, even in the absence of visual information.
But how does the brain generate these persistent internal representations? New research from Vivek Jayaraman, Shaul Druckmann and collaborators at the Janelia Research Campus in Ashburn, Virginia, outlines a potential mechanism. The findings show that in fruit flies, an internal sense of direction is maintained by what’s known as a ring attractor network.
In ring attractor networks, nodes are represented as being arranged in a ring. Neighboring nodes typically excite each other, whereas distant nodes inhibit each other, either directly or indirectly. This arrangement of excitatory and inhibitory connections constrains the network’s dynamics — a single ‘bump’ of activity moves continuously in either direction around the ring.
Neuroscientists have long theorized that ring attractors might underlie the sense of spatial orientation — Bruce McNaughton and collaborators suggested in the 1990s that head-direction cells in mammals function like ring attractors. But the new findings, published in two papers in May, provide the most direct evidence to date. “You can really see how the circuit is doing the computation,” says Larry Abbott, a theoretical neuroscientist at Columbia University who was not involved in the studies.
A network that serves as a mental compass requires a certain set of functions. It must maintain a unique representation — unless you’re lost, you generally have a sense of where you are. It also needs to represent direction continuously. If you’re walking northward and decide to reverse direction, you can’t instantaneously head south. You have to turn, transitioning through northwest, west and southwest. And the network has to remember your orientation, even in the absence of visual input. In short, a direction system requires a network that can produce strong, consistent activity that moves smoothly in either direction in the absence of visual cues. Ring attractor networks fulfill all these requirements.
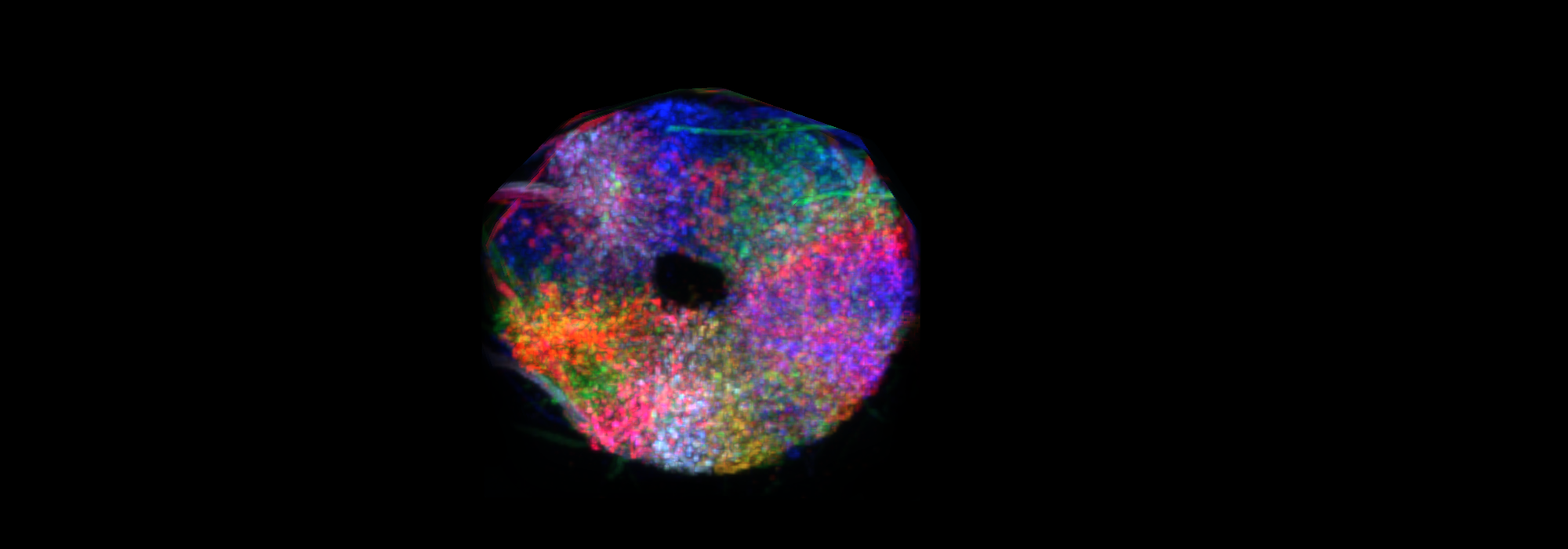
In 2015, Jayaraman and collaborator Johannes Seelig identified a compass network in fruit flies — a ring-shaped structure in the fly brain known as the ellipsoid body. They showed that the animal’s angular orientation is represented by a bump of neural activity within the ring. As the animal changes its orientation, the activity bump moves around the ring. “Activity is localized to one part of the doughnut and moves from slice to slice as the animal turns,” Jayaraman says. “That was the first clear signal that there was a ring attractor at work.”
By clicking to watch this video, you agree to our privacy policy.
The simple elegance of the structure awed neuroscientists. “The anatomy is spectacular,” Abbott says. “When you study biology, every once in a while you come across something so beautiful.”
The discovery was also striking for how well it matched theories developed through studies of rodents. “The system looks just like cartoon models that we have to explain ring attractors,” says James Knierim, a neuroscientist at Johns Hopkins University in Baltimore who was not involved in the study. “It was surprising and exciting that it was laid out this way.” Knierim and others have found indirect evidence for ring attractors in rodents but don’t yet have direct proof. For example, scientists don’t have definitive evidence of where potential ring attractor cells reside in the rat brain.
In a new study published in Science, Jayaraman and collaborators dug deeper into how the structure works. Sung Soo Kim, a researcher in Jayaraman’s lab, showed that the ring can’t represent north and southwest at the same time — the structure dampens secondary bursts of activity. Flies began the experiment with one bump of activity in the compass ring representing their heading. A second, artificial bump triggered with optogenetics quelled the activity of the original bump. “We showed directly for the first time how specific properties of the network support uniqueness of the head representation,” Jayaraman says. “I think it’s the clearest, cleanest example that we have of a neural ring attractor.”
The researchers worked with theorists Hervé Rouault and Druckmann to better understand the parameters of the network. Ring attractor networks employ both excitatory and inhibitory connections. Excitatory connections strengthen the activity of nodes with similar tuning — north-tuned nodes would most strongly excite northwest and northeast nodes. Individual nodes also inhibit other nodes — a northward node might inhibit the southward one, for example. This inhibition can take different forms. With broad uniform inhibition, activity at an individual node inhibits all other nodes equally. Alternatively, if the strength of connections follows a cosine rule, an individual node most strongly inhibits the node 180 degrees away, with progressively weaker inhibition at closer nodes and then excitation at the closest ones. Both of these options create a network where a bump of activity moves around on a circle, like a compass needle, Jayaraman says.
To distinguish between these two possibilities, Kim tested how much laser power he needed to dampen an existing bump of activity. If the network had cosine tuning, where individual units most strongly inhibit those directly opposed, it would take more power to trigger a northern bump that replaced an existing southern one. But that didn’t happen. An equivalent burst of laser power could replace one bump with another whether the two bumps were 90 degrees apart or 180. “Inhibition of the network seemed uniform,” Jayaraman says. “It’s kind of an easy structure for biology to come up with; everything is inhibited by everything else.”
By clicking to watch this video, you agree to our privacy policy.
In a second paper, published in eLife, the researchers deciphered how the activity bump moves around the ring. (A similar independent study was published in Nature at the same time.) Dan Turner-Evans and Stephanie Wegener in Jayaraman’s lab used calcium imaging and electrophysiology to map neural activity in flies whose heads were fixed in place but could still turn in different directions. The researchers focused on two structures, the ellipsoid body and a second structure called the protocerebral bridge, which resembles the handlebars on a bicycle.
Compass neurons get information from the ellipsoid body and transfer it to the protocerebral bridge. Jayaraman’s team identified a corresponding group of neurons — let’s call them turning neurons — that gets information from the bridge and sends it to the ellipsoid body. The turning neurons send their information to a segment of the ellipsoid ring just to the left or right of the slice that sent them information. Northward compass neurons, for example, would send information to two sets of turning neurons, one on each side of the bridge, which would in turn transmit to northwest or northeast compass neurons.

Credit: Stephanie Wegener
The findings provide a rare instance of nature closely matching the model that scientists developed to explain the phenomenon. “This particular network structure, this topology, is almost exactly what Bruce McNaughton’s group proposed to explain head-direction cells in mammals in 1995,” Jayaraman says. “These perfect-looking designs are like a theorist’s dream.” Abbott concurs. “Theorists often build models that are more elegant than nature, which is often kind of messy,” he says. “But this is a case where nature is neat.”
The researchers are now tracing the wiring of these structures using electron microscopy to determine whether the real anatomy matches the connectivity gleaned from activity mapping. They will also examine how animals use information from the compass. “Can we link it to an action the animal makes? What happens when we perturb the compass? Can we understand how an internal representation impacts behaviors?” Jayaraman says. He speculates that flies use this system when they are drawn to something by a strong impetus, such as the odor of food. “That’s when we think the fly will use the system to orient,” he says. “We will give the fly a goal and ask how the compass works in that case.”
In Rings, Topology Matters
| Although ring attractor networks are often visualized as a circle, the network doesn’t need to be physically arranged that way. “It’s not the topography of the network, it’s the topology,” Jayaraman says. In other words, it’s the connections that matter, rather than the physical location of the cells. “It just so happens that in the fly, topography and topology match; the units are literally arranged next to each other,” he says. He speculates that wiring optimality — the brain’s drive to be as efficient as possible when laying down neural wiring — might generate this kind of network structure. |
The findings should provide insight into mammalian compass systems as well. Scientists already knew that head-direction cells in rodents appear to employ a ring attractor network, though the mammalian versions probably don’t have the same simple doughnut shape that they have in flies. (See sidebar: In Rings, Topology Matters.) The simple fly system will help researchers dissect the more complex mammalian systems. “I think they are going to be able to figure out the circuitry of the ring attractor in ways that will give us clues for how to test things in the rat brain that we can’t test otherwise,” Knierim says. “It’s a great example of how doing research on different model systems can lend great insight into mammalian brains.”
The findings could also help researchers understand how the brain integrates information, a ubiquitous and essential computation in neural systems. “This is a circuit where we can get at the nuts and bolts of integration,” Abbott says. While the specifics of the fly navigation system won’t be exactly the same as those of other integration systems, it will reveal underlying principles, he says.


