Visualizing Apoptosis
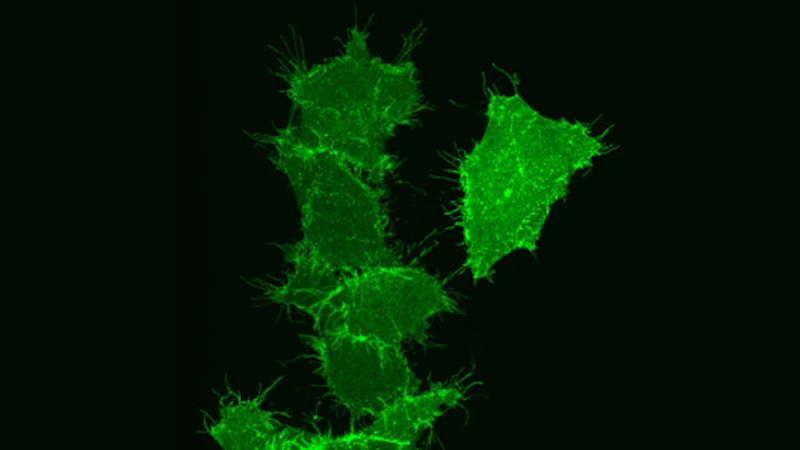
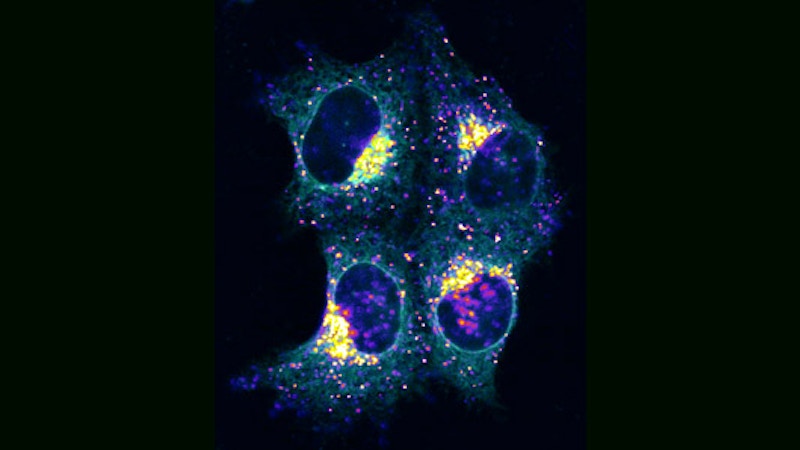
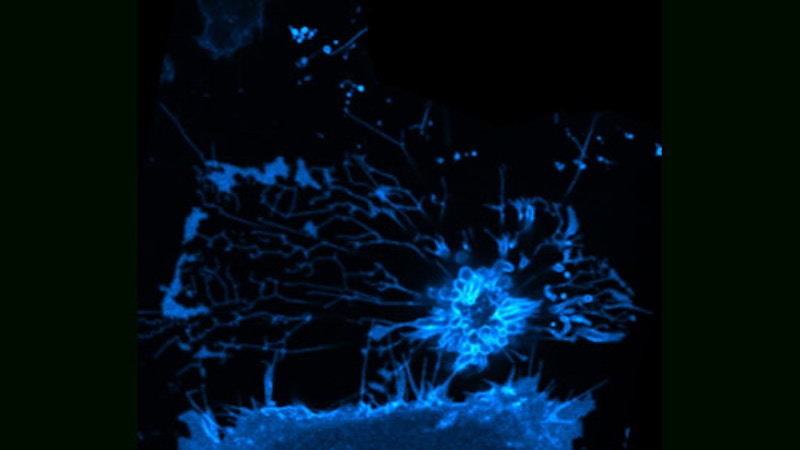
These pictures, from Dr. Roland Eils of the University of Heidelberg and the German Cancer Research Center, illustrate the process of programmed cell death, called “apoptosis,” using fluorescence microscopy. Dr. Eils’ work combines mathematical modeling with experiments in molecular cell biology to yield a detailed, quantitative understanding of basic cellular mechanisms. His knowledge in the fields of physics, mathematics and biology enables scientific results not likely attainable through a traditional approach. Such an integration of expertise comprises the relatively new field of systems biology, an illustration of this report’s emphasis on cross-disciplinary activities.
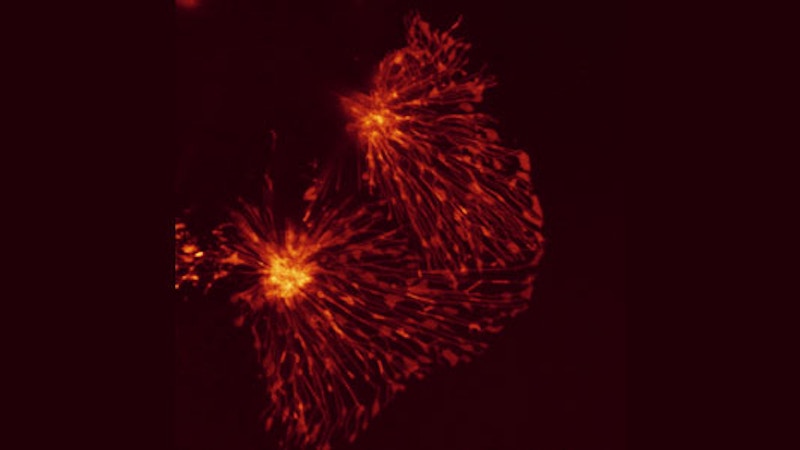
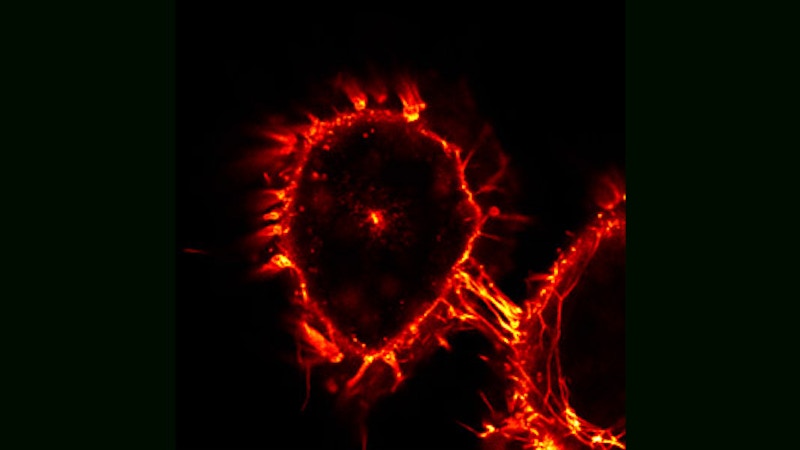
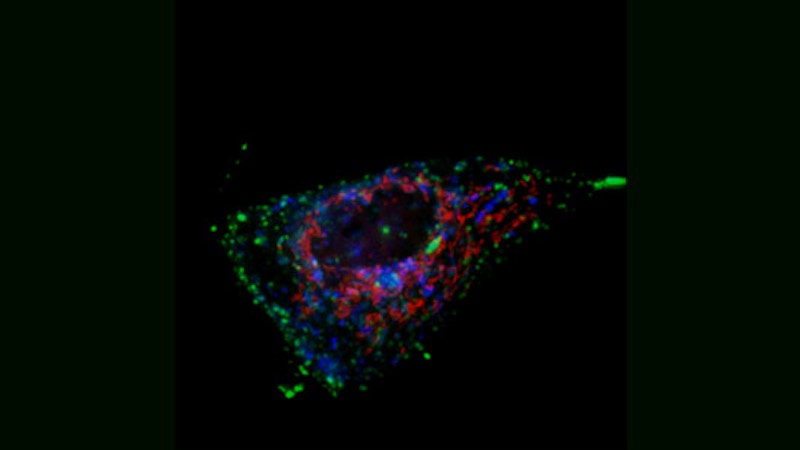
Topology of Cell Death
Here, two HeLa cells, derived from cervical cancer, are shown that have been triggered for cell death by an extracellular soluble CD95 death ligand. As soon as the CD95 death receptor is stimulated, cells start the process of apoptosis. Typically cells die within a few hours depending on the strength of cell death induction. As the cells die, they become rounder, while still keeping membrane contacts on the substrate.