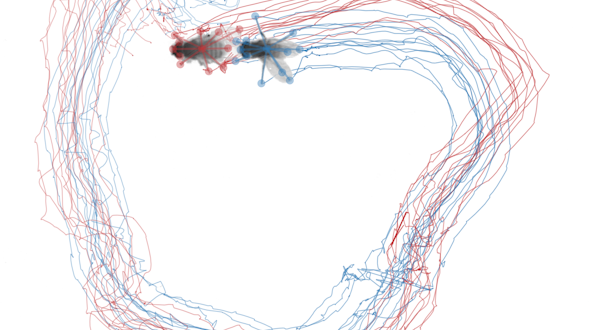
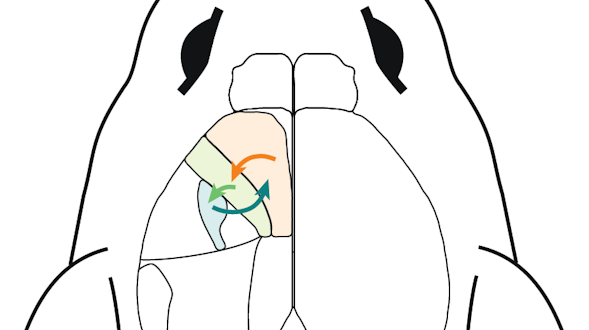
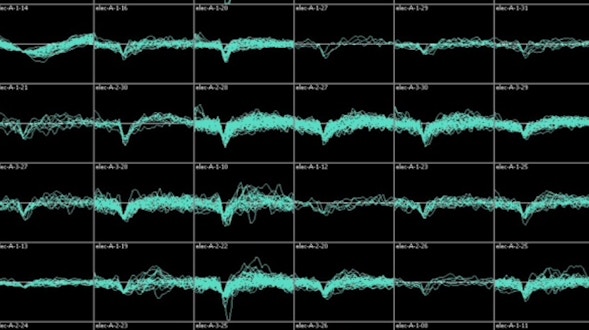
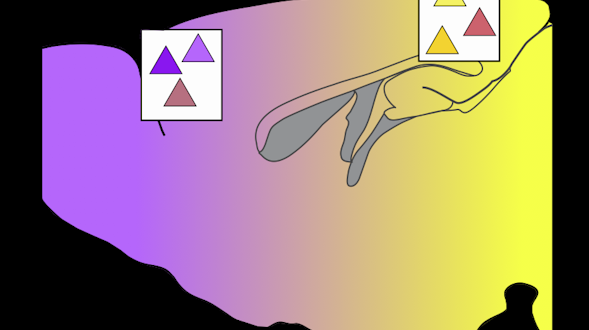
Communication is a fundamental aspect of life among animals, and yet we lack a comprehensive understanding of the underlying neural mechanisms. During communication, brains process sensory cues of different modalities, integrate those cues over time relative to changing internal states, and continually adjust ongoing actions. Here, we propose an ambitious, collaborative project to examine how the complete wiring diagram of a brain gives rise to brainwide patterns of activity that shape a social interaction, from sensation to action. In particular, we will leverage a novel connectomic resource and functional imaging methods uniquely available for Drosophila, combined with recent breakthroughs in deep learning and statistical methods for analyzing neural and behavioral data, to investigate how a female fly dynamically communicates with her social partner during courtship.
Projects
Analyzing a complex motor act at the mesoscopic scale
Michael Long, New York University
Liam Paninski, Columbia University
Attentional modulation of neuronal variability constrains circuit models
Marlene Cohen, University of Pittsburgh
Brent Doiron, University of Pittsburgh
Catching fireflies: Dynamic neural computations for foraging
Dora Angelaki, Baylor College of Medicine
Xaq Pitkow, Baylor College of Medicine
Circuit mechanisms of social cognitive computations
Winrich Freiwald, The Rockefeller University
Stefano Fusi, Columbia University
Liam Paninski, Columbia University
Computational principles of mechanisms underlying cognitive functions
Stefano Fusi, Columbia University
Daniel Salzman, Columbia University
Cortical mechanisms of multistable perception
Anthony Movshon, New York University
Corticocortical signaling between populations of neurons
Adam Kohn, Albert Einstein College of Medicine
Byron Yu, Carnegie Mellon University
Decoding internal state to predict behavior
Bernardo Sabatini, Harvard Medical School
Ryan Adams, Harvard University
Sandeep Datta, Harvard Medical School
Dense Mapping of Dynamics onto Structure in the C. elegans Brain
Manuel Zimmer, IMP Research Institute of Molecular Pathology GmbH
Dynamics of neural circuits, representation of information, and behavior
Shaul Druckmann, Howard Hughes Medical Institute, Janelia Farm Research Campus
Karel Svoboda, Howard Hughes Medical Institute, Janelia Farm Research Campus
Global brain states and local cortical processing
Matteo Carandini, University College London
Kenneth Harris, University College London
Hidden states and functional subdivisions of fronto-parietal network
Roozbeh Kiani, New York University
Higher-Level Olfactory Processing
Larry Abbott, Columbia University
How the basal ganglia forces cortex to do what it wants
Joseph Paton, Fundação D. Anna de Sommer Champalimaud e Dr. Carlos Montez Champalimaud
Brian Lau, Institut National de la Santé et de la Recherche Médicale
Interaction of sensory signals and internal dynamics during decision-making
Anne Churchland, Cold Spring Harbor Laboratory
Large-scale cortical and thalamic networks that guide oculomotor decisions
Bijan Pesaran, New York University
Xiao-Jing Wang, New York University
Large-Scale Data and Computational Framework for Circuit Investigation
E.J. Chichilnisky, Stanford University
David Brainard, University of Pennsylvania
Fred Rieke, University of Washington
Brian Wandell, Stanford University
Mechanisms of Context-Dependent Neural Integration and Short-Term Memory
Mark Goldman, University of California, Davis
Emre Aksay, Joan & Sanford I. Weill Medical College of Cornell University
Modulating the dynamics of human-level neuronal object representations
James DiCarlo, Massachusetts Institute of Technology
Network mechanisms for correcting error in high-order cortical regions
Lisa Giocomo, Stanford University
Surya Ganguli, Stanford University
Network properties and plasticity in high- and low-gain cortical states
Michael Stryker, University of California, San Francisco
Steven Zucker, Yale University
Neural Circuit Dynamics During Cognition
David Tank, Princeton University
Neural coding and dynamics for short-term memory
Ila Fiete, University of Texas at Austin
Neural computation of innate defensive behavioral decisions
Markus Meister, California Institute of Technology
David Anderson, California Institute of Technology
Pietro Perona, California Institute of Technology
Neural dynamics for a cognitive map in the macaque brain
Elizabeth A. Buffalo, University of Washington
Neural encoding and decoding of policy uncertainty in the frontal cortex
Zachary Mainen, Fundação D. Anna de Sommer Champalimaud e Dr. Carlos Montez Champalimaud
Alexandre Pouget, University of Geneva
Population dynamics across pairs of cortical areas in learning and behavior
Jonathan Pillow, The University of Texas at Austin
Spencer Smith, The University of North Carolina at Chapel Hill
Probing recurrent dynamics in prefrontal cortex
Valerio Mante, University of Zurich
William Newsome, Stanford University
Relating dynamic cognitive variables to neural population activity
Carlos Brody, Princeton University
Jonathan Pillow, The University of Texas at Austin
Maneesh Sahani, University College London
Searching for universality in the state space of neural networks
William Bialek, Princeton University
Single-trial visual familiarity memory acquisition
Nicole Rust, University of Pennsylvania
Eero P. Simoncelli, New York University
Spatiotemporal structure of neural population dynamics in the motor system
John Cunningham, Columbia University
Larry Abbott, Columbia University
Mark Churchland, Columbia University
Liam Paninski, Columbia University
The latent dynamical structure of mental activity
Maneesh Sahani, University College London
The neural basis of Bayesian sensorimotor integration
Mehrdad Jazayeri, Massachusetts Institute of Technology
The neural substrates of memories and decisions
Loren Frank, University of California, San Francisco
Uri Eden, Boston University
Towards a theory of multi-neuronal dimensionality, dynamics and measurement
Surya Ganguli, Stanford University
Krishna Shenoy, Stanford University
Toward Computational Neuroscience of Global Brains
Haim Sompolinsky, Harvard University
Florian Engert, Harvard University
Daniel Lee, University of Pennsylvania
Understanding neural computations across the global brain
Misha Ahrens, Howard Hughes Medical Institute, Janelia Farm Research Campus
Larry Abbott, Columbia University
John Cunningham, Columbia University
Jeremy Freeman, Howard Hughes Medical Institute, Janelia Farm Research Campus
Liam Paninski, Columbia University
Understanding visual object coding in the macaque brain
Doris Tsao, California Institute of Technology
Whole brain calcium imaging in freely behaving nematodes
Andrew Leifer, Princeton University