The Surprising Connection Between Hormones and Decision-Making

From a young age, Carla Golden was fascinated by the relationship between the body’s physiology and brain function. Specifically, she was interested in hormones. These chemical substances — estrogen and testosterone being two well-known examples — course through our tissues and blood vessels at levels that can vary by the day or even the hour. Hormone levels can also vary between men and women, and research has shown that these differences could play a key role in certain behavioral differences between the sexes. As she grew older, Golden wondered whether she could identify any underlying brain mechanisms behind this connection.
Today, Golden works primarily with rats to help her answer this question. While rats are not a perfect analog for humans, this well-studied animal model can still yield useful insights into human brain structure and behavior.
Golden earned her bachelor’s degree in biology with a concentration in neuroscience from the Schreyer Honors College at Pennsylvania State University, and a Ph.D. in neuroscience from the Icahn School of Medicine at Mount Sinai. A second-year Junior Fellow with the Simons Society of Fellows, she is now completing postdoctoral training in the Constantinople Lab at New York University (NYU). We recently discussed her work. Our conversation has been edited for clarity.
In your doctoral work at Mount Sinai, you focused on autism. What did that research teach you about brain differences between the sexes?
Data has shown that boys are more likely to be diagnosed with autism than girls, but the reasons for this remain unclear. The disparity could be because boys are more often referred for autism diagnoses than girls, or because girls are better able to mask signs of autism in social settings. There is a lot of active research focused on identifying the underlying reasons for this difference.
At Mount Sinai, one of my experiments explored these questions through a rat model of fragile X syndrome, a genetic disorder that causes delayed speech and deep difficulties with attention and focus. The syndrome, so-called because it is caused by a mutation on the X chromosome, is the most common cause of inherited autism. Since males have one X chromosome and females have two, males are diagnosed with fragile X more often. Unlike females, males don’t have that second X chromosome to mask the fragile X mutation.
Can you tell us more about that experiment?
The experiment examined the behavior of several varieties of male and female rats. Some male rats had the fragile X mutation on their X chromosome, while some female rats had the mutation on either one or both of their X chromosomes. Another subset of male and female rats had no fragile X mutations at all. These rats served as controls in our study.
We gave all rats a task that required their attention: poking their nose through a hole to receive sugar water. We gradually decreased the amount of time the rats had to poke the screen, from 60 to 2.5 seconds. The shorter times demanded more attention; they had less available time to complete this task.
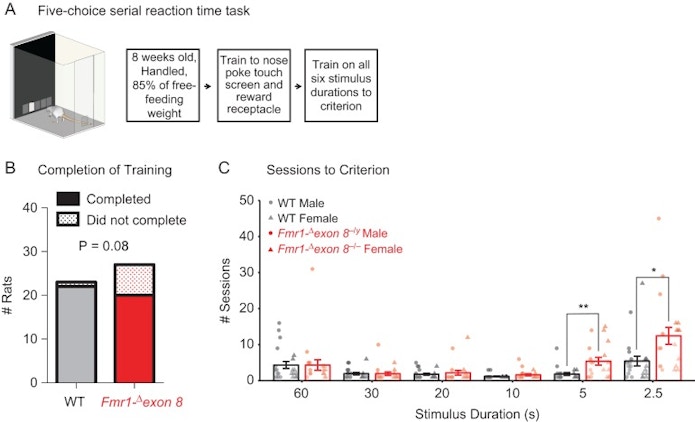
We found that the male rats with the fragile X mutation, and female rats with the mutation on both X chromosomes, were equally bad at completing this task. It appeared that being female in and of itself offered no protective benefit, at least in these conditions. We also completed a structural analysis of the animals’ brain function — down to the connections between neurons — that led to these attention deficits. Not only did we publish the results of the initial experiment in the journal Cerebral Cortex and of the structural analysis in the journal Translational Psychiatry, but this work has since inspired my interest in higher-order cognitive functioning in autism as a way to explore decision-making in general.
Now you’ve expanded your interests. What is the current focus of your work at NYU?
An important part of making a decision is learning which actions yield the best results. Say you’re driving to get groceries and deciding between turning left or right at an intersection. You know from experience that if you turn left, you get to a farmers’ market. If you turn right, you will get to a big grocery store. In this scenario, experience has taught you the pros and cons of each option, and you make your choice accordingly. People with certain psychiatric conditions often have difficulty in these types of situations.
With my work, I’d like to understand what happens in the brain as we learn how to make these choices, and the brain processes that exist to make us better or worse at this type of learning. Like my previous work, I use rats as a model for study. However, there are some challenges with using animal models for studies of decision-making. Unlike with people, we can’t ask the rats why they made one choice versus the other. We can only go by their actions. Their decision to go left versus right, for example, when both options are equally possible, can tell us which choice they thought was best.
In our experiment, rats get a certain amount of water if they wait for an unpredictable amount of time. If at any time they decide that they are sick of waiting, they have the option of moving on to another, potentially larger, amount of water. We’re interested in tracking which rats wait, and what amount of water is worth that wait. We also want to know how making changes to the amount of water they get after waiting may also lead them to make different choices.
Specifically, I’m looking at how the hormone estradiol, a type of estrogen that is present in greater quantities in females than in males, could impact rats’ abilities to learn the consequences of their choices and apply that knowledge to making future decisions. Higher levels of estradiol are linked to higher levels of dopamine, a brain chemical tied to learning and reward.
What have you discovered so far?
The work is ongoing, so we will make more discoveries as we continue, but some interesting trends are emerging. Female rats have an estrous cycle, analogous to the human menstrual cycle, during which levels of hormones like estradiol and progesterone wax and wane.
The animals’ estrous cycle runs every four to five days. During the ovulation portion of the cycle, known as estrus, hormone levels surge. We’ve also seen that, during estrus, rats are much faster at learning when it is worth their effort. If during estrus they deduce that they will only get a little water no matter how hard they work, the rats take longer between trials, and generally seem bored. And if they deduce that they will be able to get the maximum reward, they work as fast as possible to do so.
Preliminary research in rats also suggests that dopamine also surges during estrus. We’re still doing work to confirm this, but it appears more dopamine remaining in synapses [the connection points between neurons] may impact the rats’ ability to learn.
Finally, what are your thoughts about being a Simons Junior Fellow?
I greatly appreciate the intergenerational aspect of the Junior Fellowship. We have Senior Fellows — these are some of the most eminent researchers in their fields — who give insightful and historical perspectives about how scientific understanding within a given area has changed over time, or how experimental techniques have evolved. I am very grateful for these insights.
For example, one expert in the field of decision-making told me about a collection of experiments in the 1960s. Even though the experiment used a comparatively crude measure of motivation that did not involve decision-making, it still showed how estrogen could change female motivation. These foundational studies helped me focus my own work with today’s cutting-edge tools, without repeating the important work of the scientists who came before me.


